-
Novo Nordisk Introduces Ozempic Pill; Available in the US Q2 2026
Novo Nordisk Introduces Ozempic Pill; Available in the US Q2 2026
2026-02-08 -
Watching the Super Bowl? Doctors Warn About Hidden Health Risks for Fans
Watching the Super Bowl? Doctors Warn About Hidden Health Risks for Fans
2026-02-08 -
Male-to-Female Ratio for Autism Has Decreased Over Time
Male-to-Female Ratio for Autism Has Decreased Over Time
2026-02-08
New Products
Changzhou Extraordinary Pharmatech co., LTD.
Changzhou Extraordinary Pharmatech co., Mg, grams, kilograms, tonnage pharmaceutical intermediates, customized production;Sales of various chemical test and agent catalysts.
Company has strong ability of research and development, has multi-function production workshop, at the same time with high temperature and high pressure, temperature, such as small and medium-sized reactor,And equipped with a gas chromatograph (GC), high performance liquid chromatograph (HPLC) analysis of testing instruments, such as,Meet from mg、kg、ton identifying and customization of rare compounds screening, can accept FTE, CRO the custom synthesis,process optimization, route design, technology outsourcing services,with the domestic many scientific research institutions and r&d team to develop, in the chemical industry park and dozens of chemical plants have long-term cooperative relations, and in hubei, jiangxi, shandong has a processing base, provides the guarantee to provide a steady stream of supply of goods.
We have our own factories and laboratories.We focus on technology and services.I hope we have the opportunity to cooperation.
We will give you the best price and service.
Welcome to the advisory prices.

 Deutsche
Deutsche Español
Español français
français italiano
italiano português
português 日本語
日本語 한국어
한국어 العربية
العربية русский
русский bahasa Indonesia
bahasa Indonesia Tiếng Việt
Tiếng Việt







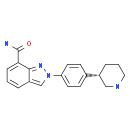 MK-4827 (HCl)
MK-4827 (HCl) Niraparib
Niraparib (R)-tert-butyl ...
(R)-tert-butyl ... (R)-tert-butyl ...
(R)-tert-butyl ...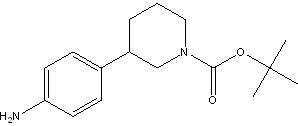 tert-butyl 3-(4...
tert-butyl 3-(4...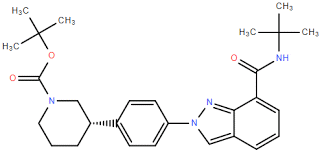 (3S)-3-[4-[7-[[...
(3S)-3-[4-[7-[[...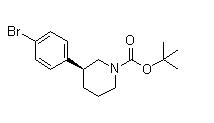 (3S)-3-(4-Bromo...
(3S)-3-(4-Bromo...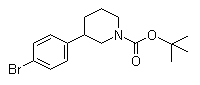 3-(4-Bromopheny...
3-(4-Bromopheny...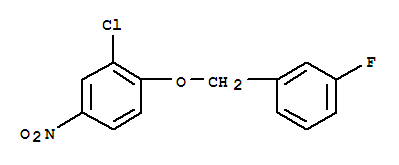 3-Chloro-4-(3-f...
3-Chloro-4-(3-f...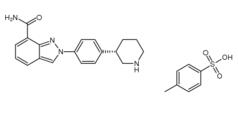 Niraparib p-tol...
Niraparib p-tol...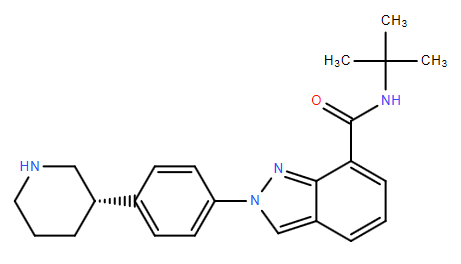 N-(1,1-Dimethyl...
N-(1,1-Dimethyl...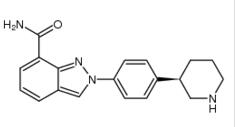 2-{4-[(3S)-pipe...
2-{4-[(3S)-pipe...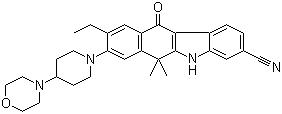 Alectinib
Alectinib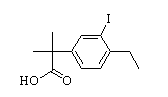 2-(4-ethyl-3-io...
2-(4-ethyl-3-io...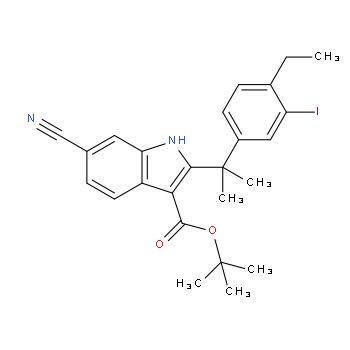 tert-butyl 6-cy...
tert-butyl 6-cy...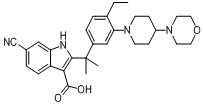 6-cyano-2-(2-(4...
6-cyano-2-(2-(4...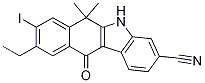 9-ethyl-8-iodo-...
9-ethyl-8-iodo-...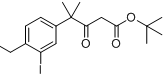 tert-butyl 4-(4...
tert-butyl 4-(4...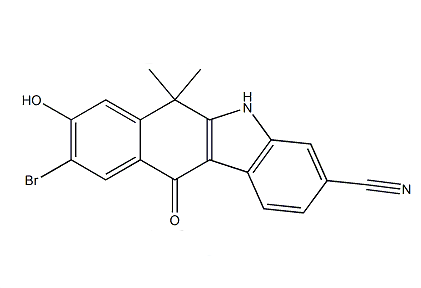 9-broMo-8-hydro...
9-broMo-8-hydro...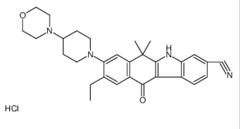 9-ethyl-6,6-diM...
9-ethyl-6,6-diM...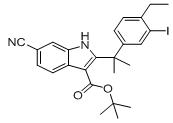 tert-butyl 6-cy...
tert-butyl 6-cy...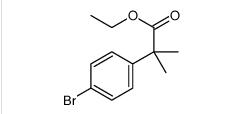 ethyl 2-(4-broM...
ethyl 2-(4-broM...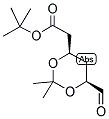 ert-Butyl (4R-c...
ert-Butyl (4R-c...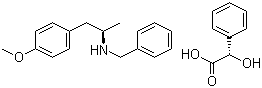 (2S)-Hydroxy(ph...
(2S)-Hydroxy(ph...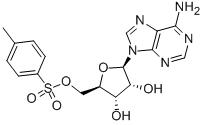 5-Tosyladenosin
5-Tosyladenosin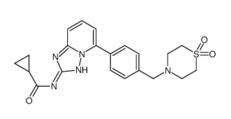 Filgotinib
Filgotinib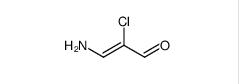 3-amino-2-chlor...
3-amino-2-chlor...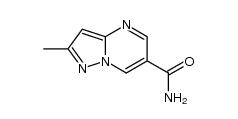 2-Methylpyrazol...
2-Methylpyrazol...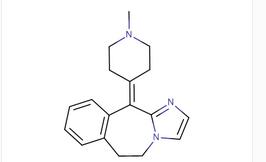 11-(1-Methylpip...
11-(1-Methylpip...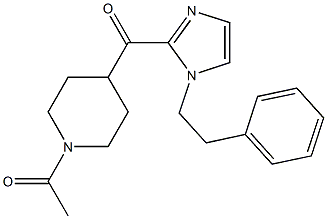 1-(4-(1-PHENETH...
1-(4-(1-PHENETH...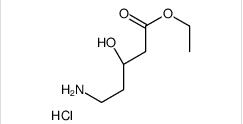 ethyl (3R)-5-am...
ethyl (3R)-5-am...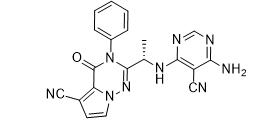 LAS191954 free ...
LAS191954 free ...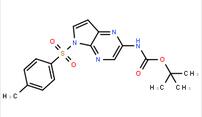 tert-butyl 5-to...
tert-butyl 5-to...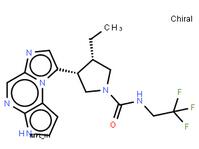 ABT-494 Interme...
ABT-494 Interme...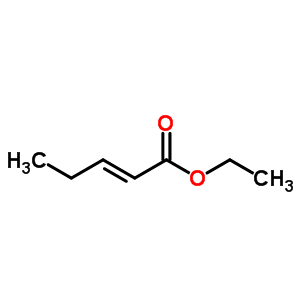 ethyl (2E)-pent...
ethyl (2E)-pent...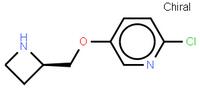 abt594 Intermed...
abt594 Intermed...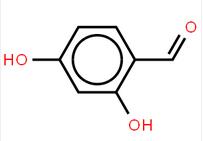 LOXO101 Interme...
LOXO101 Interme...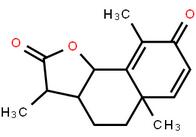 LOXO101 Interme...
LOXO101 Interme...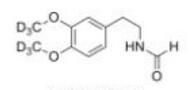 Deutetrabenazin...
Deutetrabenazin...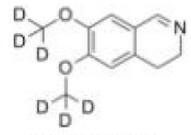 Deutetrabenazin...
Deutetrabenazin...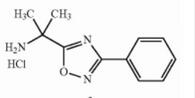 Naldemedine tos...
Naldemedine tos...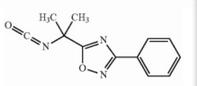 Naldemedine tos...
Naldemedine tos...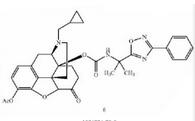 Naldemedine
Naldemedine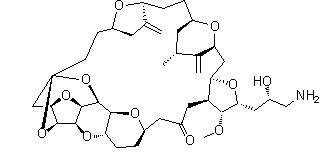 Eribulin
Eribulin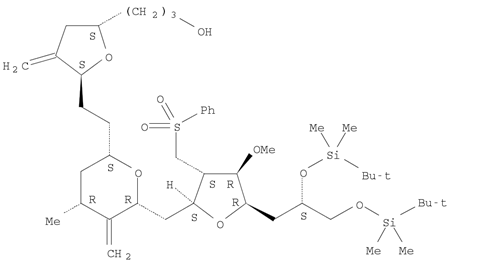 2-Furanpropanol...
2-Furanpropanol...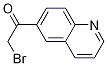 2-BroMo-1-quino...
2-BroMo-1-quino...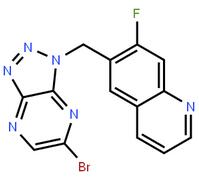 6-[(6-Bromo-1H-...
6-[(6-Bromo-1H-...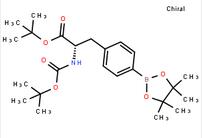 tert-butyl (S)-...
tert-butyl (S)-...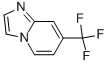 7-Trifluorometh...
7-Trifluorometh...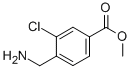 methyl 4-(amino...
methyl 4-(amino...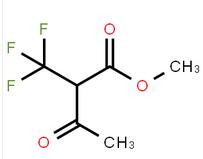 methyl 3-oxo-2-...
methyl 3-oxo-2-...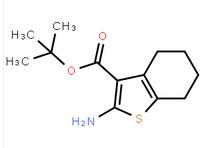 2-AMino-4,5,6,7...
2-AMino-4,5,6,7...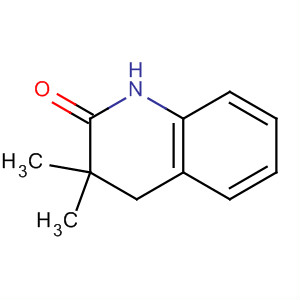 2(1H)-Quinolino...
2(1H)-Quinolino...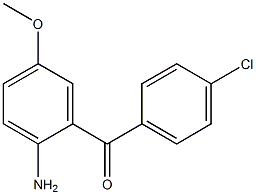 Methanone, (2-a...
Methanone, (2-a...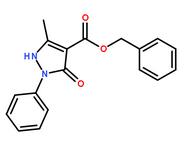 benzyl 5-methyl...
benzyl 5-methyl...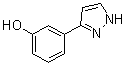 3-(1H-pyrazol-5...
3-(1H-pyrazol-5...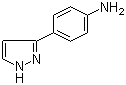 4-(1H-Pyrazol-3...
4-(1H-Pyrazol-3...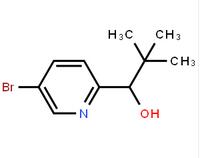 1-(5-bromo-pyri...
1-(5-bromo-pyri...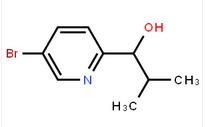 1-(5-bromo-pyri...
1-(5-bromo-pyri...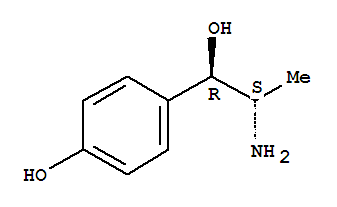 Benzenemethanol...
Benzenemethanol...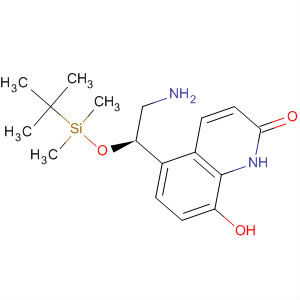 2(1H)-Quinolino...
2(1H)-Quinolino...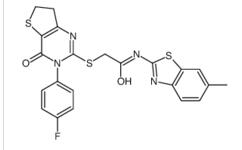 2-{[3-(4-Fluoro...
2-{[3-(4-Fluoro...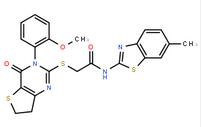 2-((3-(2-methox...
2-((3-(2-methox...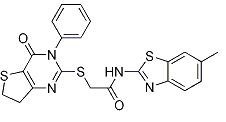 N-(6-Methyl-2-b...
N-(6-Methyl-2-b...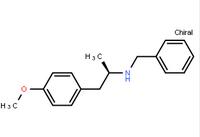 (R)-(-)-1-(4-me...
(R)-(-)-1-(4-me...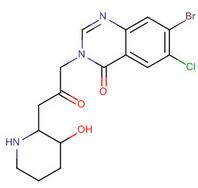 4(3H)-Quinazoli...
4(3H)-Quinazoli...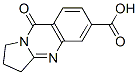 9-OXO-1,2,3,9-T...
9-OXO-1,2,3,9-T...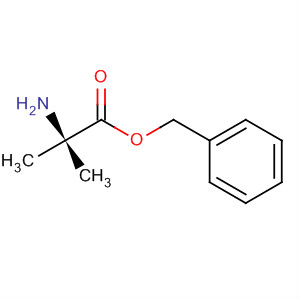 Alanine, 2-meth...
Alanine, 2-meth...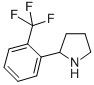 2-(2-TRIFLUOROM...
2-(2-TRIFLUOROM...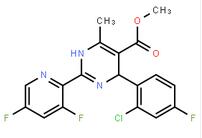 (-)-4(R)-(2-Chl...
(-)-4(R)-(2-Chl...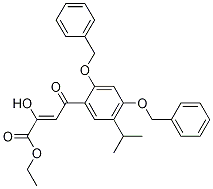 2-Butenoic acid...
2-Butenoic acid...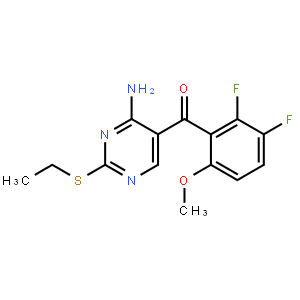 [4-amino-2-(eth...
[4-amino-2-(eth...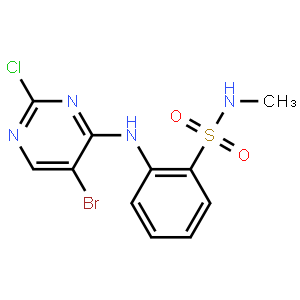 Benzenesulfonam...
Benzenesulfonam...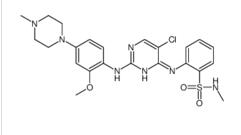 ALK inhibitor 2
ALK inhibitor 2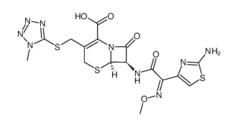 Cefmenoxime hyd...
Cefmenoxime hyd...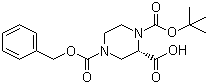 (S)-N-1-Boc-N-4...
(S)-N-1-Boc-N-4...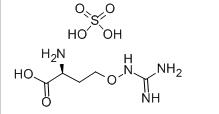 L-CANAVANINE SU...
L-CANAVANINE SU...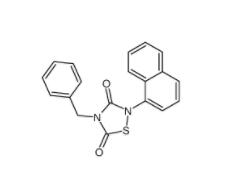 Tideglusib 865...
Tideglusib 865...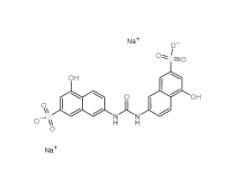 Disodium 7,7-(c...
Disodium 7,7-(c...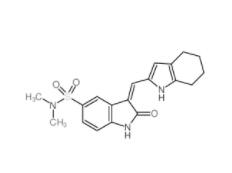 SU 6656 330161...
SU 6656 330161...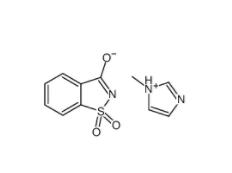 Saccharin 1-met...
Saccharin 1-met...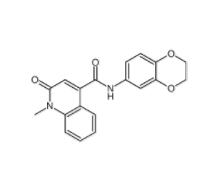 CeMMEC13 17908...
CeMMEC13 17908...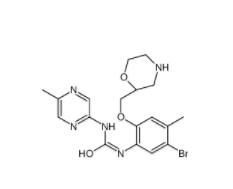 Rabusertib 911...
Rabusertib 911...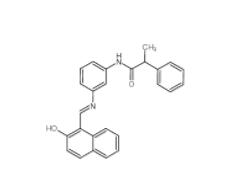 Salermide 1105...
Salermide 1105...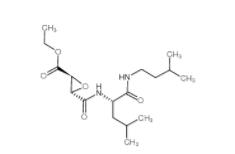 EST 88321-09-9
EST 88321-09-9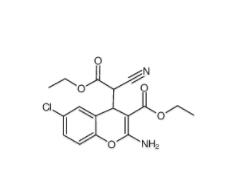 SC79 305834-79...
SC79 305834-79...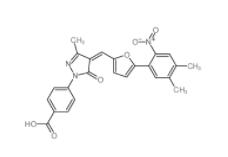 C646 328968-36...
C646 328968-36...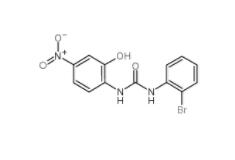 1-(2-bromopheny...
1-(2-bromopheny... Dp44mT 152095-...
Dp44mT 152095-... Deguelin 522-1...
Deguelin 522-1... PD168393 19442...
PD168393 19442... YO01027 209984...
YO01027 209984...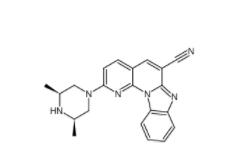 DC10539 182235...
DC10539 182235...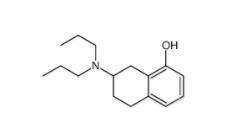 8-OH-DPAT 7895...
8-OH-DPAT 7895...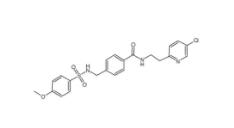 YU238259 19437...
YU238259 19437...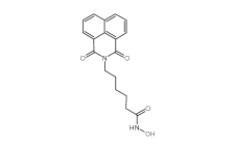 Scriptaid 2873...
Scriptaid 2873...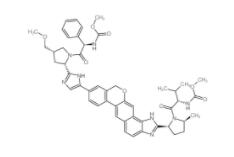 Velpatasvir 13...
Velpatasvir 13...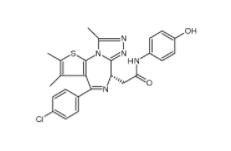 OTX015 202590-...
OTX015 202590-...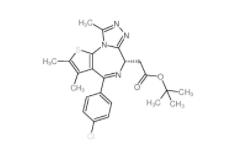 (+)-JQ-1 12685...
(+)-JQ-1 12685...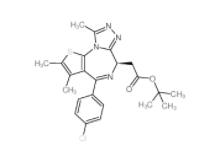 (-)-JQ-1 12685...
(-)-JQ-1 12685...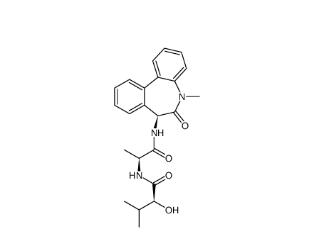 LY 900009 2099...
LY 900009 2099...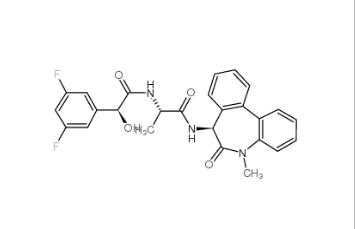 LY-411575 2099...
LY-411575 2099...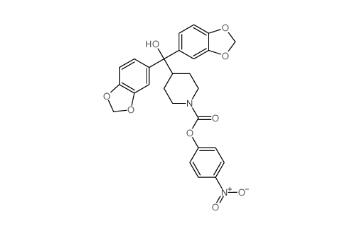 (4-nitrophenyl)...
(4-nitrophenyl)...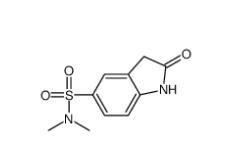 N,N-dimethyl-2-...
N,N-dimethyl-2-...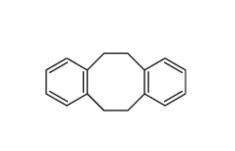 5,6,11,12-tetra...
5,6,11,12-tetra...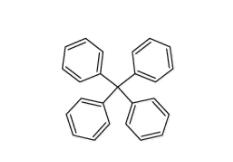 Tetraphenylmeth...
Tetraphenylmeth...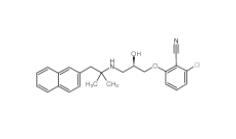 2-chloro-6-[(2R...
2-chloro-6-[(2R... 1616380-54-1
1616380-54-1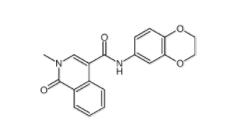 N-(2,3-dihydrob...
N-(2,3-dihydrob...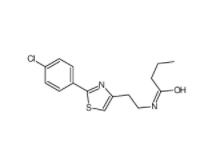 N-[2-[2-(4-chlo...
N-[2-[2-(4-chlo...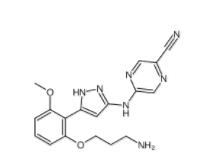 5-(5-(2-(3-amin...
5-(5-(2-(3-amin...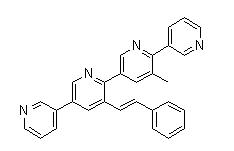 3-Methyl-3-[(1E...
3-Methyl-3-[(1E...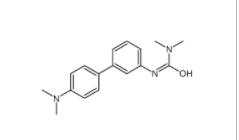 3-[4-(Dimethyla...
3-[4-(Dimethyla...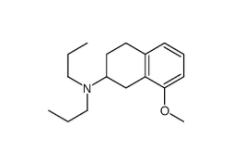 8-Methoxy-N,N-d...
8-Methoxy-N,N-d...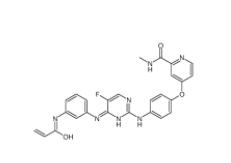 4-{4-[(4-{[3-(A...
4-{4-[(4-{[3-(A...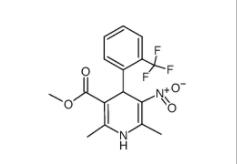 1,4-Dihydro-2,6...
1,4-Dihydro-2,6...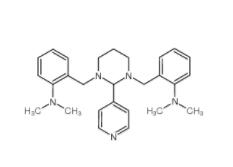 2-[[3-[[2-(dime...
2-[[3-[[2-(dime...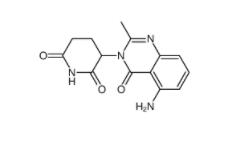 CC-122 1015474...
CC-122 1015474...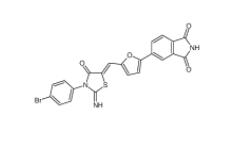 Bioymifi 14200...
Bioymifi 14200...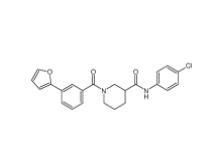 N-(4-chlorophen...
N-(4-chlorophen...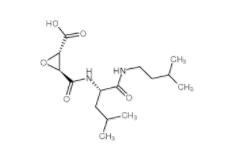 E-64C 76684-89...
E-64C 76684-89...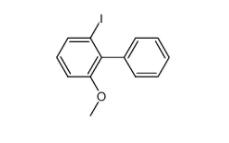 2-iodo-6-methox...
2-iodo-6-methox... 4EP-Directory l...
4EP-Directory l...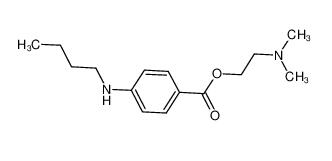 tetracaine 94-...
tetracaine 94-...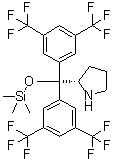 (S)-2-[Bis[3,5-...
(S)-2-[Bis[3,5-...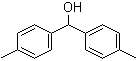 Bis(4-methylphe...
Bis(4-methylphe...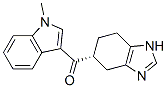 (1-Methyl-1H-In...
(1-Methyl-1H-In...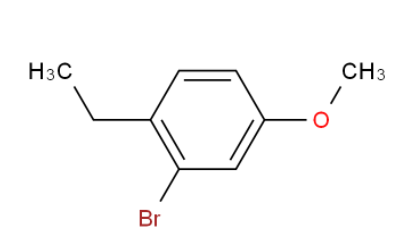 2-Bromo-1-ethyl...
2-Bromo-1-ethyl...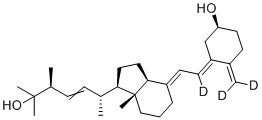 25-Hydroxy vita...
25-Hydroxy vita...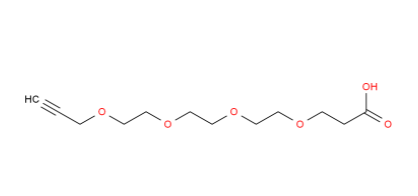 Propargyl-PEG4-...
Propargyl-PEG4-... 1EP-Directory l...
1EP-Directory l...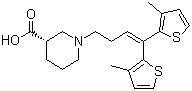 Tiagabine 1151...
Tiagabine 1151...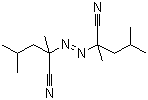 2,2-Azobis(2,4-...
2,2-Azobis(2,4-...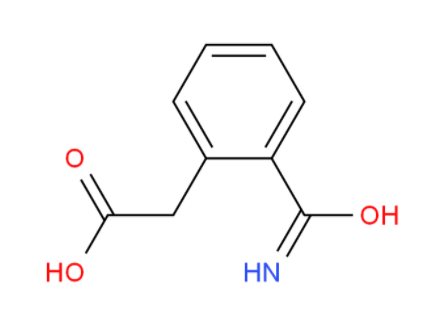 [2-(aminocarbon...
[2-(aminocarbon...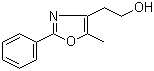 2-(5-Methyl-2-p...
2-(5-Methyl-2-p...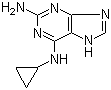 2-Amino-6-cyclo...
2-Amino-6-cyclo...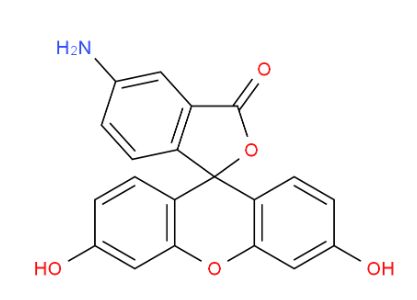 MOLPORT-004-153...
MOLPORT-004-153...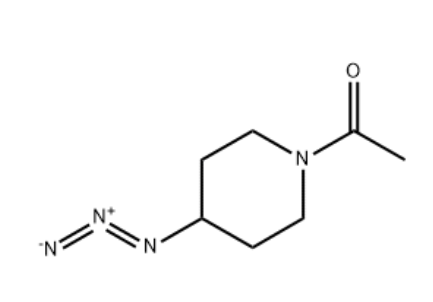 1-(4-Azidopiper...
1-(4-Azidopiper...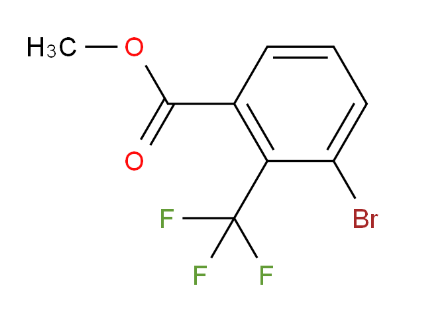 METHYL 3-BROMO-...
METHYL 3-BROMO-... 2EP-Directory l...
2EP-Directory l... 3EP-Directory l...
3EP-Directory l...



